The Study
This comprehensive review, synthesizing evidence from 2015 to 2024, delves into the multifaceted nature of Autism Spectrum Disorder (ASD), a heterogeneous, lifelong neurodevelopmental condition. Researchers Nating Xiong and Zhikang Yu from Meizhou People’s Hospital examined a broad spectrum of areas including epidemiology, clinical diagnosis, neurobiology, genetics, environmental risk factors, mortality, and treatment. The review aims to integrate the latest scientific understanding, particularly in the genomic era, to provide a holistic view of ASD’s etiology and potential therapeutic avenues. It highlights how diagnostic expansion and methodological advances have led to increased prevalence estimates and a deeper appreciation of the condition’s phenotypic, etiological, and outcome heterogeneity.
The authors specifically focused on robust genetic contributions, the interaction of prenatal and perinatal environmental exposures with genetic susceptibility, and neuroimaging insights into altered brain development. They also addressed the significant issue of increased mortality in individuals with ASD and evaluated current treatment modalities, including the role of Applied Behavior Analysis (ABA), alongside emerging genomic technologies like CRISPR-based approaches for precision therapies.
Key Findings
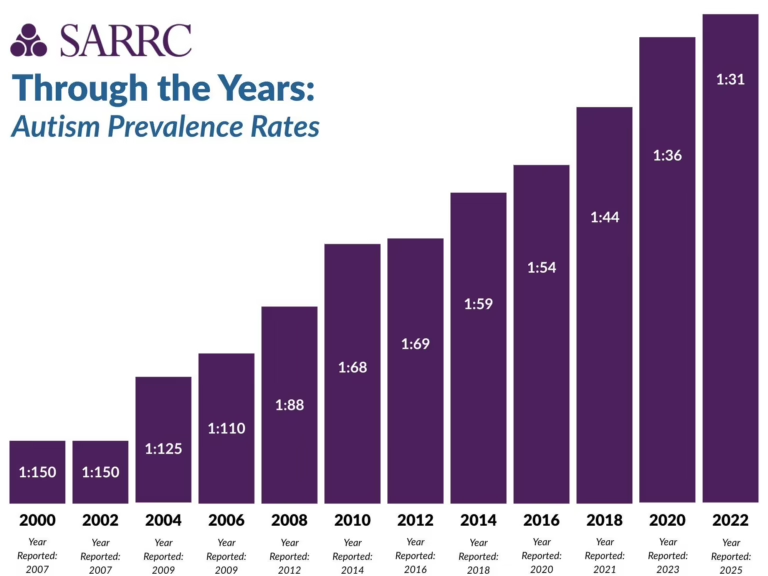
The review underscores the significant increase in ASD prevalence, with recent US Centers for Disease Control and Prevention (CDC) data indicating approximately 1 in 31 children in the United States are affected, a substantial rise from earlier estimates of 2–4 cases per 10,000 children in the 1960s. Globally, about 1 in 127 individuals across all ages are estimated to be on the autism spectrum. ASD is nearly four times more common in males than females, a disparity partly explained by the “female protective effect” hypothesis, suggesting females require a higher genetic burden for clinical presentation.
Genetic factors are identified as the largest contributors to ASD risk, with twin studies showing 80–90% concordance in monozygotic twins. Both common inherited variants (e.g., GRIN2B, AVPR1A, OXTR, 5-HTTLPR, EN2, GABR3) and rare de novo or inherited mutations (e.g., SHANK3, NRXN1, FOXP2, CDH8, SYNGAP1, and copy number variants or CNVs) are implicated. These genetic disruptions often converge on pathways critical for synaptic function, chromatin regulation, and neurodevelopment.
Environmental exposures also play a crucial role, interacting with genetic susceptibility. Well-supported risk factors include advanced parental age, maternal infection/fever during pregnancy (associated with a ~1.3-fold increased risk), preterm birth, and obstetric complications. A growing body of evidence links prenatal exposure to ambient air pollution (e.g., PM2.5, heavy metals) and certain pesticides to increased ASD risk, often through mechanisms involving oxidative stress, immune dysregulation, and epigenetic changes.
Neuroimaging studies reveal altered cortical developmental trajectories and atypical large-scale network connectivity (e.g., default mode, salience, and sensory integration networks) associated with core ASD symptoms and comorbidities. Individuals with ASD face substantially elevated premature mortality, with overall standardized mortality ratios (SMRs) of ~2–3 times higher than the general population, rising to >3–5 times higher for those with co-occurring intellectual disability. Leading causes include epilepsy (affecting 20–30% of autistic individuals), medical comorbidities, and external causes like accidental injury (notably drowning) and suicide.
Diagnosis relies on clinical assessment based on DSM-5-TR criteria, with early developmental screening recommended between 18–24 months. Gold standard diagnostic tools include the Autism Diagnostic Observation Schedule, Second Edition (ADOS-2) and the Autism Diagnostic Interview–Revised (ADI-R). Genetic testing, such as chromosomal microarray analysis (CMA) and whole-exome sequencing (WES), is increasingly used for etiological clarification, offering diagnostic yields of 10–20% and 30–40% respectively. Neuroimaging and metabolic screening are used selectively.
Current treatments are primarily symptomatic, with early, intensive, and individualized behavioral interventions providing the greatest functional benefit. Applied Behavior Analysis (ABA) is highlighted as the most empirically substantiated modality, with models like Pivotal Response Treatment (PRT) and Discrete Trial Training (DTT) demonstrating strong effects on communication, social-adaptive functioning, and early cognitive outcomes. The Early Start Denver Model (ESDM) integrates ABA principles with developmental neuroscience for toddlers. Pharmacotherapy, such as risperidone for irritability, targets associated behavioral challenges.
Emerging genomic technologies, particularly CRISPR-based gene editing, offer powerful experimental models and hold potential for precision therapies for selected monogenic or high-impact CNV-associated forms of ASD. While preclinical successes exist, significant challenges remain regarding safety, efficient brain-wide delivery, off-target effects, and ethical considerations, meaning no CRISPR-based therapy has entered human trials as of 2024.
Clinical Implications
For practicing BCBAs and RBTs, these findings reinforce the critical importance of early and intensive behavioral interventions, particularly ABA-derived models, as the cornerstone of effective treatment. The detailed understanding of genetic and environmental factors can inform discussions with families about ASD etiology and the potential benefits of genetic testing for diagnostic clarification and future precision therapies. Clinic owners and researchers should note the emphasis on individualized, functionally oriented treatment paradigms and the need for interdisciplinary collaboration to address the complex needs and comorbidities, including the elevated mortality risk. This research also provides a strong evidence base for advocating with insurers for comprehensive coverage of intensive ABA services, given its empirically substantiated benefits on functional outcomes. Furthermore, the insights into the female protective effect highlight the need for increased awareness and improved diagnostic practices to identify ASD in females who may present with more subtle or camouflaged symptoms.
Fast Facts
| Key Point | Why It Matters for ABA |
|---|---|
| ASD prevalence: 1 in 31 US children | Highlights the widespread need for effective ABA services and early intervention. |
| ABA is most empirically substantiated modality | Reinforces ABA’s central role and supports advocacy for its funding and implementation. |
| Increased mortality in ASD, especially with ID | Emphasizes the need for comprehensive care, including comorbidity management and safety planning within ABA programs. |
| CRISPR shows future promise for monogenic ASD | Signals potential for highly individualized, gene-targeted therapies, though still preclinical, influencing long-term research directions. |
Expert Perspective
The genomic era is ushering in a new understanding of ASD’s complex origins, reinforcing the need for early, individualized behavioral interventions while paving the way for future precision therapies.
Source: dovepress.com